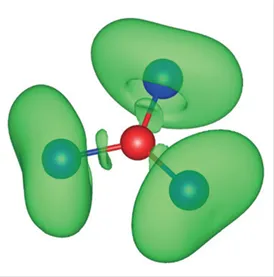
Carbonate build up by metals: Whereas alkali metal carbonates are mostly insoluble in liquid ammonia, a carbonate analogue is formed by Bi and Sn by reaction of deeply colored solutions of polyanionc bismuthides and stannides in liquid ammonia and can be isolated as the potassium salt K5SnBi3⋅9 NH3. In contrast to carbonate, the highly charged [BiSn3]5− ion decomposes readily in the presence of traces of water.
For article see: DOI:10.1002/anie.201709700 or DOI:10.1002/ange.201709700
K. Mayer, J. V. Dums, W. Klein, T. F. Fässler [SnBi3]5– − A Carbonate Analogue Comprising Exclusively Metal Atoms
Angew. Chem. Int. Ed. 56 (2017) 15159–15163