Development of a multiplex algae toxin immunoassay for the monitoring of algal blooms in surface water
OBJECTIVE
The online monitoring system is designed to collect information on certain parameters that can lead to excessive growth of cyanobacteria and to evaluate them online using an algorithm. These include nutrient concentrations of nitrate, phosphate, and oxygen as well as pH, solids content and redox potential. In addition to the parameters mentioned, it should also be possible to monitor the concentration of the free and intracellular toxins.
Cyanotoxins are classified based on their mode of action in invertebrates. Distinctions are made between hepatotoxins, neurotoxins, and dermatotoxins, among others. This project focuses on the hepatotoxin microcystin-LR and the neurotoxins anatoxin a and saxitoxin. These toxins are predominantly intracellular toxins, which are released into water after cell death and subsequent lysis.
- Immunoassay for the simultaneous detection of different cyanotoxins
- Sample preparation including separation of free and cell bound cyanotoxins,
preconcentration and elimination of matrix
- Algae reactor for testing the sensor-technology provided by the project partners
- Field tests at the Easter Lakes near to Iffeldorf
METHODS
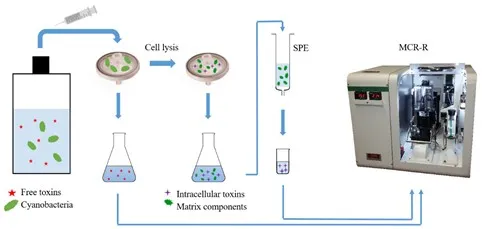
The assay, used for the detection of cyanotoxins is a regenerative indirect competitive flow through immunoassay microarray, which is performed with the microarray chip reader designed for research (MCR-R). The flow cell of the microchip is formed by a surface-modified glass slide with covalently bound cyanotoxins and a PMMA-single-channel carrier with inlet and outlet holes.
For sample preparation, a glass fiber filter or membrane filter is used to separate free toxins from the cyanobacteria. After filtration, the free cyanotoxins in the eluate are determined directly using the MCR-R. The cyanobacteria are lysed by repeated freezing and thawing of the filter and extracted with a suitable solvent. Then the toxins can be separated from the matrix by solid phase extraction or immunomagnetic separation and preconcentrated at the same time. Subsequently, the originally intracellular toxins are determined with the MCR-R.
INVOLVED PHD STUDENTS
M. Sc. Andreas Auernhammer
PARTNERS
A.U.G. Signals Ltd., Toronto
Hydroisotop GmbH, Schweitenkirchen
FINANCIAL SUPPORT
AIF-ZIM (ZF4810901LL9)