New Publication in Biosensors
J. Neumair, M. Kröger, E. Stütz, C. Jerin, A. M. Chaker, C. B. Schmidt-Weber and M. Seidel
Biosensors (2023)
Abstract
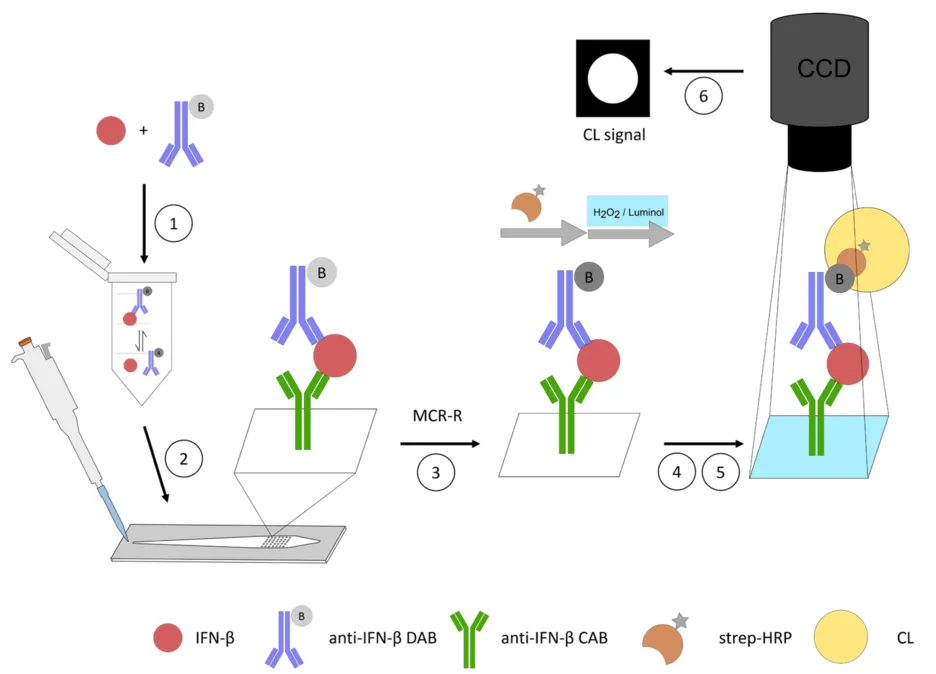
Protein biomarkers in nasal secretions can be used as a measure to differentiate between allergies, airway diseases and infections for non-invasive diagnostics. The point-of-care quantification of biomarker levels using flow-based microarray facilitates precise and rapid diagnosis and displays the potential for targeted and effective treatment. For the first time, we developed a flow-based chemiluminescence sandwich microarray immunoassay (CL-SMIA) for the quantification of nasal interferon-beta (IFN-β) on the Microarray Chip Reader-Research (MCR-R). Polycarbonate foils are used as a cost-effective surface for immobilizing capture antibodies. By using a commercially available set of anti-human IFN-β antibodies, the CL-SMIA can be compared directly to an enzyme-linked immunosorbent assay (ELISA) performed in microtiter plates concerning the bioanalytical performance and economic issues. Pre-incubation of the sample with detection antibodies facilitates the lower consumption of detection antibodies, as this allows for a longer interaction time between the antibody and the biomarker. The direct injection of pre-incubated samples into the microarray chips eliminates the adsorption of proteins in the tubing as well as the contamination of the tubing and valves of the MCR-R with clinical samples. The small flow cell allows for a low sample volume of 50 μL. The limit of detection of 4.53 pg mL−1 was slightly increased compared to a sandwich ELISA performed on microtiter plates which were 1.60 pg mL−1. The possibility to perform the CL-SMIA in a multiplexed mode makes it a promising assay for the rapid and cost-effective non-invasive detection of biomarkers in nasal secretions.