Internally Mixed Soot Aerosols
Objectives
- Characterization of soot internally mixed with metal compounds
- Correlation between oxidation reactivity, soot structure, metal species and metal content
- Prove applicability of commonly-used thermo-optical techniques for quantification of organic carbon (OC) and elemental carbon (EC)
Methods of Approach
- Produce surrogate iron containing soot
- Analyze synthetical and real soot using Raman-Microscopy, Temperature-Programmed-Oxidation (TPO) and Photoacoustic spectroscopy among other techniques
Description
Combustion generated soot particles are hazardous environmental pollutants, especially concerning human health and climate. Primary aims of the project are to synthesize multicomponent internally mixed soot particles and to develop a method for characterization of organic and inorganic composition, soot structure and oxidation reactivity of synthetic and real soot internally mixed with metal compounds. Those metal compounds may catalyse the oxidation of soot particles, leading to significant changes in physical, thermochemical and optical properties.
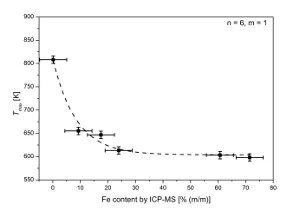
So far, iron-containing soot was produced by adding iron pentacarbonyle to a propane/air diffusion flame. Among various other techniques, the soot is analyzed by Raman Microspectroscopy for soot structure analysis and Temperature-Programmed Oxidation for characterization of the thermo-chemical properties. It could be shown, that soot structure does not change significantly by varying the quantity of iron internally mixed with the soot. In contrary, soot oxidation is severely impacted as the emission of CO and CO2 during the TPO experiment shifted towards lower temperature compared to undoped soot. The temperature of maximum emission Tmax is used as criterion for soot reactivity. It decreases drastically with increasing Fe content in soot and thus soot reactivity is increased substantially with increasing Fe content. Furthermore, soot with certain Fe content could be completely burnt in inert atmosphere, proving that commonly-used thermo-optical techniques for quantification of OC and EC are not applicable if inorganic material is present in the aerosol sample to be analyzed.
The study will be extended from iron-containing soot to soot internally mixed with various inorganic salts.
Pubilcations
H. Bladt, N. P. Ivleva & R. Niessner, Internally Mixed Multicomponent Soot: Impact of Different Salts on Soot Structure and Thermo-chemical Properties, Aerosol Science and Technology 2014, 70, 26-35
J. Moldanova, E. Fridell, H. Winnes, S. Holmin-Fridell, J. Bolnan, A. Jedyuska, V. Tishkova, B. Demirdjian, S. Joulie, H. Bladt, N. Ivleva & R. Niessner, Physical and Chemical Characterisation of PM Emissions from Two Ships Operating in European Emission Control Areas, Aerosol Measurement Technology Discussion, 2013, 6, 3931-3982
O. Popovicheva, E. Kireeva, N. Persiantseva, M. Timofeev, H. Bladt, N. P. Ivleva, R. Niessner, J. Moldanova, Microscopic characterization of individual particles from multicomponent ship exhaust, Journal of Environmental Monitoring 2012, 14, 3101-3110
H. Bladt, J. Schmid, E. D. Kireeva, O. B. Popovicheva, N. M. Perseantseva, M. A. Timofeev, K. Heister, J. Uihlein, N. P. Ivleva, R. Niessner, Impact of Fe content in laboratory-produced soot aerosol on its composition, structure and thermo-chemical properties, Aerosol Science and Technology 2012, 46, 1337-1348.